
Practical communication tools for transfer
How to take the most of communication to facilitate the arrival of innovative solutions on the market? Tips and tools at the XVII B&L.

How to take the most of communication to facilitate the arrival of innovative solutions on the market? Tips and tools at the XVII B&L.
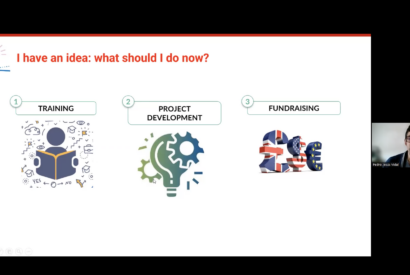
Biocat's innovation team has shared some funding sources and acceleration programs of Catalan, national and European scope for researchers.

Do you want to know ways-to-do, tools and success stories to include the gender perspective in research and transfer? Some keys at the B&L.

Do you want to learn about mechanisms to boost the transfer of your research results? Biocat shares useful tools at the 15th B&L.

What is knowledge transfer? Learn about the step-by-step path to transferring solutions and the Network's support tools to make it possible.

More than 80 people attended the first face-to-face session of the TECSAM Network Kick-Off Meeting, a meeting point for member groups.

Do you want to learn about tools to transfer research results to the market? Discover how the TECSAM Network helps member groups in tech transfer.

Do you need funding to bring your solution to the market? At the 13th B&L, Patricio Hunt at Intelectium shares the 6 keys to attract private investors.
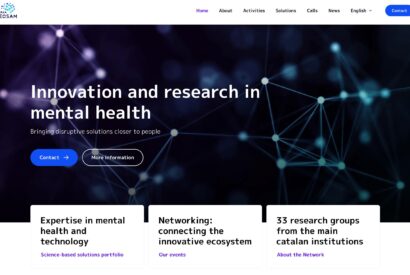
The TECSAM Network's new website is now available! Solutions portfolio, events, training capsules and more, just a click away. Dive into the world of innovation in mental health!

Looking for funding to develop and bring your research project to market? On 14 December we will be holding the 13th Breakfast&Learn, where we will share the steps to follow to access private funding. Conference by Intelectium. Sign up!
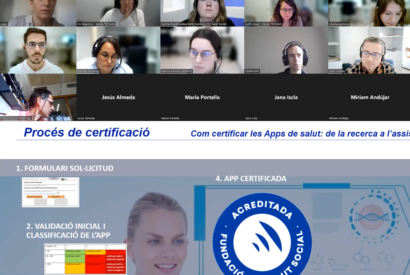
Certification of health apps: ethical principles, privacy and accessibility are some of the key aspects. This was one of the central reflections of the 12th B&L by TIC Salud Social, the official accreditation body in Catalonia. Now available in video!

On Wednesday 2 November at 9am, the TECSAM Network is holding the XII Breakfast&Learn, entitled “how to accredit or certify health Apps: from research to care” where Carme Pratpadua and Bufill from TIC Social Health will talk about the certification process, the roadmap and the main obstacles for the development of health apps. Join us!